Report Overview
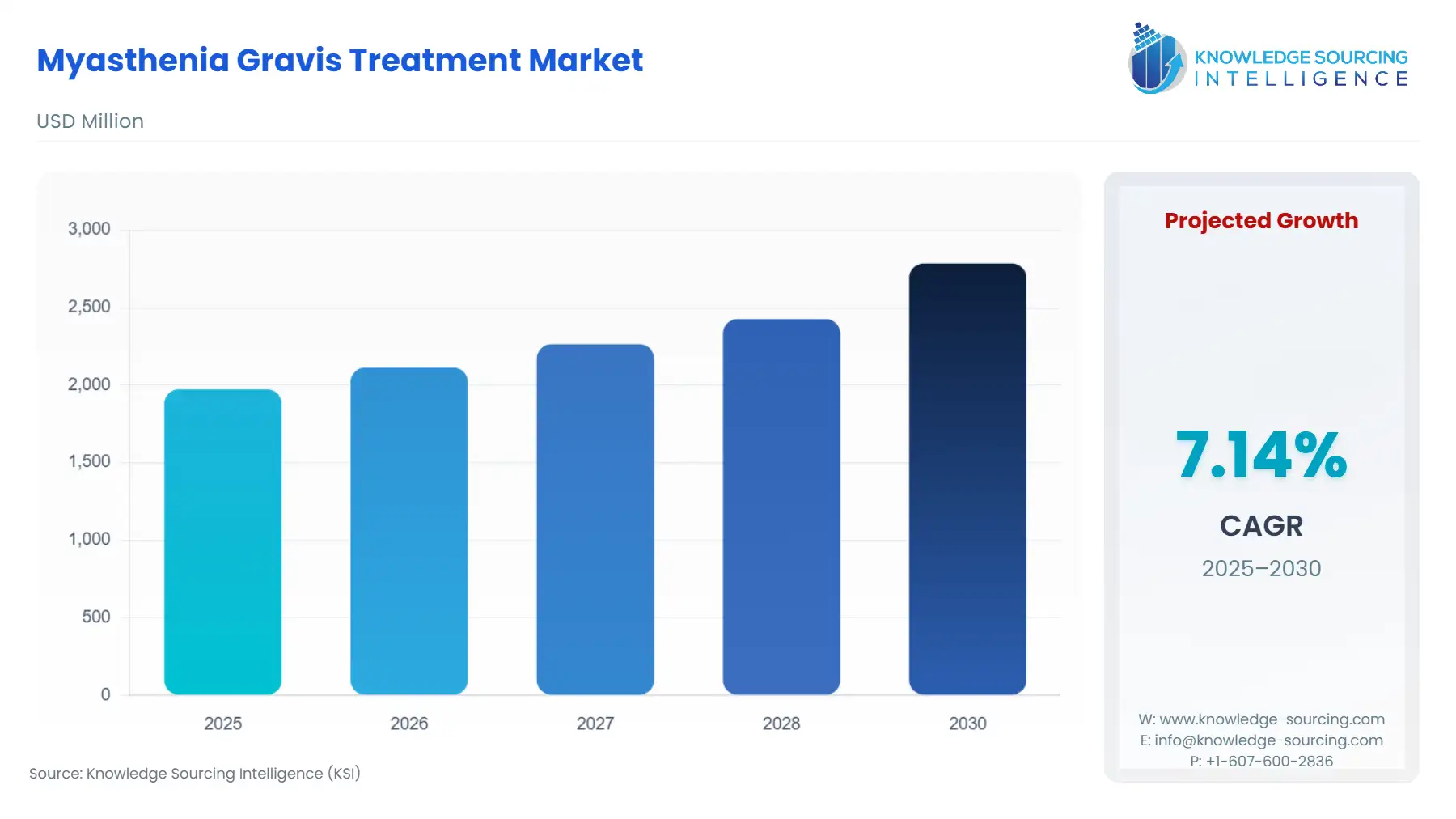
The myasthenia gravis treatment market is estimated to grow at a CAGR of 7.14%, from US$2.785 billion by 2030 to US$1.973 billion in 2025.
Myasthenia gravis is a type of chronic neuromuscular disease that generally causes weakness in the voluntary muscles. It is majorly caused by a problem with the signals sent between the nerves and the muscles. The treatment of myasthenia gravis includes various types of medications and surgeries, like blood transfusion, steroids, and muscle strengtheners.
The global myasthenia gravis treatment market is expected to witness major growth with the increasing global cases of autoimmune diseases.
Myasthenia gravis is a type of chronic autoimmune disease that affects the voluntary muscles of the body. The global cases of autoimmune diseases have witnessed significant growth in the past few years, majorly in the older or adult population. Similarly, the increasing research and development of the pharmaceutical sector is also estimated to surge market growth during the forecasted timeline.
Myasthenia Gravis Treatment Market Growth Drivers:
Growing global cases of autoimmune disorder
The growing global prevalence of autoimmune disorders is among the key factors propelling the myasthenia gravis treatment market growth during the forecasted timeline. Autoimmune disorders are conditions in which the body's immune system attacks its health tissues. Myasthenia gravis is an autoimmune disorder that destroys communication between muscles and nerves.
The global cases of autoimmune disorders witnessed a major growth during the past few years. The University of Oxford, in its report, stated that about 10% of the total global population, with 13% of women and 7% of men, is affected by the diseases.
Rising investments in the global pharmaceutical research sector
The major factor boosting the global myasthenia gravis treatment market’s expansion is the increasing global investment in the pharmaceutical sector research and development. With the growing global investment in the pharmaceutical sector, an efficient and more advanced pharmaceutical treatment process, like medication and surgery, for myasthenia gravis diseases is expected to increase. The governments of various countries, like the USA, Germany, Canada, and India, have introduced key policies and investment opportunities to boost their domestic pharmaceutical research and development.
For instance, in December 2024, the Ministry of Health and Family Welfare of the India Government allocated INR 5,000 crore for 5 years, starting from 2023-24, aiming to transform the domestic pharmaceutical-technology sector. The investment also aims to enhance the pharmaceutical sector's domestic research and development ecosystem.
Myasthenia Gravis Treatment Market Restraints:
Higher cost of treatment
The major factor challenging the global myasthenia gravis treatment market’s growth is the higher cost of treatment. The treatment cost of myasthenia gravis includes the research and development of highly precise medication and surgery processes, increasing this cost. The development process of myasthenia gravis medication includes various key procedures, like drug discovery, preclinical research, and extensive research by research institutes, and governmental agencies. With the market's extensive and expensive drug development process, the growth is expected to witness a major challenge during the forecasted timeline.
Myasthenia Gravis Treatment Market Geographical Outlook:
North America is forecasted to hold a major share of the myasthenia gravis treatment market
The North American region is expected to witness significant growth in the global myasthenia gravis treatment market, mainly due to the increasing development of pharmaceutical technologies. The growth of the pharmaceutical technology and research & development ecosystem in North America, especially in countries like the USA and Canada, witnessed significant growth. The USA is among the leading producers of pharmaceutical drugs and technologies, boosting the myasthenia gravis treatment market expansion during the estimated timelines. Similarly, introducing key governmental policies to boost pharmaceutical research and development is also expected to significantly grow the market.
Myasthenia Gravis Treatment Market Products Offered by Key Companies:
Merck KGaA is a German-based global leader in science and technology, offering expertise across multiple sectors, like life science, healthcare, and electronics. The company offers research solutions, which include a healthcare pipeline, clinical trials, a global R&D hub, and global health institutes, among others. In the healthcare sector, the company offers technological solutions and products for various categories, like fertility, neurology & immunology, oncology, cardiovascular, metabolism, & endocrinology, among others. In the global myasthenia gravis treatment market, the company offers MyClad, a type of generalized Myasthenia Gravis
AstraZeneca is a global leader in pharmaceutical and science-led biopharmaceutical development. The company is among the leading global research-based biopharmaceutical companies, providing expertise across multiple sectors like oncology, cardiovascular, renal, & metabolism, rare diseases, and vaccine & immune therapies. It also offers research and development across numerous pharmaceutical fields, including transformative science, data science & AI, clinical innovation, digital health, and precision medicine. In the global myasthenia gravis treatment market, the company announced the FDA approval of ULTOMIRIS generalized myasthenia gravis, showing early effects and improved daily living.
Myasthenia Gravis Treatment Market Key Developments:
In August 2024, Johnson & Johnson, a global pharmaceutical leader, announced that the company is seeking U.S. Food and Drug Administration (FDA) approval for its nipocalimab, designed for treating antibody-positive generalized myasthenia gravis. The drug offers multiple benefits to patients suffering from myasthenia gravis. The nipocalimab drug features a high binding affinity and specificity to the immunoglobulin G (IgG) binding site.
In February 2024, Dianthus Therapeutics, a global leader in a clinical-stage biotechnology company, launched a Phase 2 trial to test DNTH103 in generalized myasthenia gravis. The company further stated that the phase 2 trial aims to enroll up to 60 participants with positive antibodies, which target acetylcholine receptor (AChR), to further determine the efficiency of the drugs. The major aim of the phase 2 trial is to assess the safety and tolerance of the DNTH103 generalized myasthenia gravis. In this trial, the DNT103 drug is injected under the skin of the participants.
Myasthenia Gravis Treatment Market Scope:
| Report Metric | Details |
|---|---|
| Study Period | 2021 to 2031 |
| Historical Data | 2021 to 2024 |
| Base Year | 2025 |
| Forecast Period | 2026 – 2031 |
| Companies |
|
Report Metric | Details |
Myasthenia Gravis Treatment Market Size in 2025 | US$1.973 billion |
Myasthenia Gravis Treatment Market Size in 2030 | US$2.785 billion |
Growth Rate | CAGR of 7.14% |
Study Period | 2020 to 2030 |
Historical Data | 2020 to 2023 |
Base Year | 2024 |
Forecast Period | 2025 – 2030 |
Forecast Unit (Value) | USD Billion |
Segmentation |
|
Geographical Segmentation | North America, South America, Europe, Middle East and Africa, Asia Pacific |
List of Major Companies in Myasthenia Gravis Treatment Market |
|
Customization Scope | Free report customization with purchase |
The Myasthenia Gravis Treatment market is segmented and analyzed as follows:
By Application
Medication
Pyridostigmine
Cyclosporine
Azathioprine
Surgery
Other
By End User
Hospitals
Clinics
Others
By Geography
North America
USA
Canada
Mexico
South America
Brazil
Argentina
Others
Europe
Germany
Spain
United Kingdom
France
Italy
Others
Middle East and Africa
Saudi Arabia
UAE
Others
Asia Pacific
China
Japan
Australia
India
Indonesia
Thailand
Others